Also know, what does an allosteric modulator bind to?
A ligand which binds to a receptor at a site distinct from that of the endogenous agonist endogenous agonist. Orthosteric binding is mutually exclusive. The effects of an allosteric modulator are saturable – they have an upper y limit.
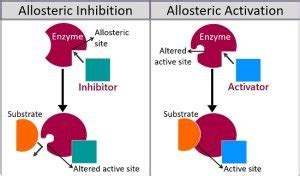
One may also ask, what is the allosteric site of an enzyme? n. The place on an enzyme where a molecule that is not a substrate may bind, thus changing the shape of the enzyme and influencing its ability to be active.
In this way, what does allosteric modulation mean?
In pharmacology and biochemistry, allosteric modulators are a group of substances that bind to a receptor to change that receptor's response to stimulus. Some of them, like benzodiazepines, are drugs. Modulators and agonists can both be called receptor ligands.
What is allosteric antagonist?
A drug that binds to a receptor at a site distinct from the active site. A non-competitive antagonist binds to an allosteric (non-agonist) site on the receptor to prevent activation of the receptor. A reversible antagonist binds non-covalently to the receptor, therefore can be “washed out”.